- Radiometric dating - Wikipedia
- What is Radioactive Dating? - Definition & Facts
- What is Radiocarbon Dating?
The stable isotopes are carbon 12 and carbon Carbon 14 is continually being formed in the upper atmosphere by the effect of cosmic ray neutrons on nitrogen 14 atoms. It is rapidly oxidized in air to form carbon dioxide and enters the global carbon cycle. Plants and animals assimilate carbon 14 from carbon dioxide throughout their lifetimes. When they die, they stop exchanging carbon with the biosphere and their carbon 14 content then starts to decrease at a rate determined by the law of radioactive decay. Radiocarbon dating is essentially a method designed to measure residual radioactivity. By knowing how much carbon 14 is left in a sample, the age of the organism when it died can be known.
It must be noted though that radiocarbon dating results indicate when the organism was alive but not when a material from that organism was used. There are three principal techniques used to measure carbon 14 content of any given sample— gas proportional counting, liquid scintillation counting, and accelerator mass spectrometry. Gas proportional counting is a conventional radiometric dating technique that counts the beta particles emitted by a given sample.
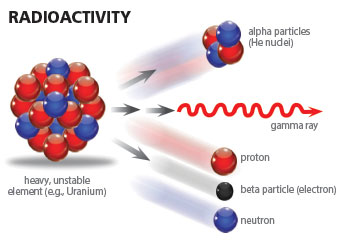
Beta particles are products of radiocarbon decay.
Radiometric dating - Wikipedia
In this method, the carbon sample is first converted to carbon dioxide gas before measurement in gas proportional counters takes place. Liquid scintillation counting is another radiocarbon dating technique that was popular in the s. In this method, the sample is in liquid form and a scintillator is added.
This scintillator produces a flash of light when it interacts with a beta particle. A vial with a sample is passed between two photomultipliers, and only when both devices register the flash of light that a count is made. Accelerator mass spectrometry AMS is a modern radiocarbon dating method that is considered to be the more efficient way to measure radiocarbon content of a sample. In this method, the carbon 14 content is directly measured relative to the carbon 12 and carbon 13 present.
In the 19th and early 20th century incredibly patient and careful archaeologists would link pottery and stone tools in different geographical areas by similarities in shape and patterning. Then, by using the idea that the styles of objects evolve, becoming increasing elaborate over time, they could place them in order relative to each other - a technique called seriation. In this way large domed tombs known as tholos or beehive tombs in Greece were thought to predate similar structures in the Scottish Island of Maeshowe. This supported the idea that the classical worlds of Greece and Rome were at the centre of all innovations.
What is Radioactive Dating? - Definition & Facts
Some of the first radiocarbon dates produced showed that the Scottish tombs were thousands of years older than those in Greece. The barbarians of the north were capable of designing complex structures similar to those in the classical world.
- How Does Carbon Dating Work?
- free cell phone hookup.
- dating someone with your same last name;
- What Is Radioactive Dating, and How Does It Work?.
- jay ramras dating.
- chennai dating girl.
- You must create an account to continue watching;
Other high profile projects include the dating of the Turin Shroud to the medieval period, the dating of the Dead Sea Scrolls to around the time of Christ, and the somewhat controversial dating of the spectacular rock art at Chauvet Cave to c. Radiocarbon dating has also been used to date the extinction of the woolly mammoth and contributed to the debate over whether modern humans and Neanderthals met.
But 14 C is not just used in dating. Using the same techniques to measure 14 C content, we can examine ocean circulation and trace the movement of drugs around the body.
But these are topics for separate articles. See more Explainer articles on The Conversation. Dealers, collectors and curators: Standing up for minorities in Egypt — York, York. An analytical journey measuring contaminants in water around the globe — Portsmouth, Hampshire. Geniuses, Heroes and Saints: Available editions United Kingdom. Rachel Wood , Australian National University. Make no bones about it, radiocarbon dating has transformed our understanding of the past.
Dating history When living things die, tissue is no longer being replaced and the radioactive decay of 14 C becomes apparent.
Carbon dioxide is used in photosynthesis by plants, and from here is passed through the food chain. Dating advances Radiocarbon dates are presented in two ways because of this complication. Establishing dates Moving away from techniques, the most exciting thing about radiocarbon is what it reveals about our past and the world we live in. Such trapped Ar is not problematical when the age of the rock is in hundreds of millions of years.
The dating equation used for K-Ar is: Some of the problems associated with K-Ar dating are Excess argon. This is only a problem when dating very young rocks or in dating whole rocks instead of mineral separates. Minerals should not contain any excess Ar because Ar should not enter the crystal structure of a mineral when it crystallizes. Thus, it always better to date minerals that have high K contents, such as sanidine or biotite. If these are not present, Plagioclase or hornblende. If none of these are present, then the only alternative is to date whole rocks. Some 40 Ar could be absorbed onto the sample surface.
This can be corrected for. Most minerals will lose Ar on heating above o C - thus metamorphism can cause a loss of Ar or a partial loss of Ar which will reset the atomic clock. If only partial loss of Ar occurs then the age determined will be in between the age of crystallization and the age of metamorphism. If complete loss of Ar occurs during metamorphism, then the date is that of the metamorphic event. The problem is that there is no way of knowing whether or not partial or complete loss of Ar has occurred. Examples of questions on this material that could be asked on an exam.
Prior to the best and most accepted age of the Earth was that proposed by Lord Kelvin based on the amount of time necessary for the Earth to cool to its present temperature from a completely liquid state. Principles of Radiometric Dating Radioactive decay is described in terms of the probability that a constituent particle of the nucleus of an atom will escape through the potential Energy barrier which bonds them to the nucleus. Thus, if we start out with 1 gram of the parent isotope, after the passage of 1 half-life there will be 0.
What is Radiocarbon Dating?
Some examples of isotope systems used to date geologic materials. To see how we actually use this information to date rocks, consider the following: To account for this, we first note that there is an isotope of Sr, 86 Sr, that is: If we divide equation 4 through by the amount of 86 Sr, then we get: Note also that equation 5 has the form of a linear equation, i.
How can we use this? In nature, however, each mineral in the rock is likely to have a different amount of 87 Rb.
- match dating costs.
- dating factory forum!
- what are the best types of rocks to use for radiometric dating;
Thus, once the rock has cooled to the point where diffusion of elements does not occur, the 87 Rb in each mineral will decay to 87 Sr, and each mineral will have a different 87 Rb and 87 Sr after passage of time. The Concordia curve can be calculated by defining the following: The discordia is often interpreted by extrapolating both ends to intersect the Concordia. Pb leakage is the most likely cause of discordant dates, since Pb will be occupying a site in the crystal that has suffered radiation damage as a result of U decay.
U would have been stable in the crystallographic site, but the site is now occupied by by Pb. An event like metamorphism could heat the crystal to the point where Pb will become mobile. Another possible scenario involves U leakage, again possibly as a result of a metamorphic event. U leakage would cause discordant points to plot above the cocordia.
The Age of the Earth A minimum age of the Earth can be obtained from the oldest known rocks on the Earth. So far, the oldest rock found is a tonalitic Gneiss metamorphic rock rock from the Northwest Territories, Canada, with an age of 3. This gives us only a minimum age of the Earth. Is it likely that we will find a rock formed on the Earth that will give us the true age of the Earth?
