- Carbon Dating: The History Of Life On Earth (Video)
- The Age of the Earth - Radiocarbom Dating as a Current Scientific Clock: Jonathan Ring
- Professor Timothy H. Heaton
- Navigation menu
Carbon and other radioisotopes are used to measure the age of fossils, rocks, and other materials that make up Earth's geologic history. These techniques are reliable and valid. They provide clues into our earliest origins. And they contribute to the hundreds of lines of evidence supporting Charles Darwin's theory of evolution , which continues to stand the test of time. To learn more, watch the video above and read the transcript below. And don't forget to leave a comment at the bottom of the page. Talk nerdy to me!
Carbon Dating: The History Of Life On Earth (Video)
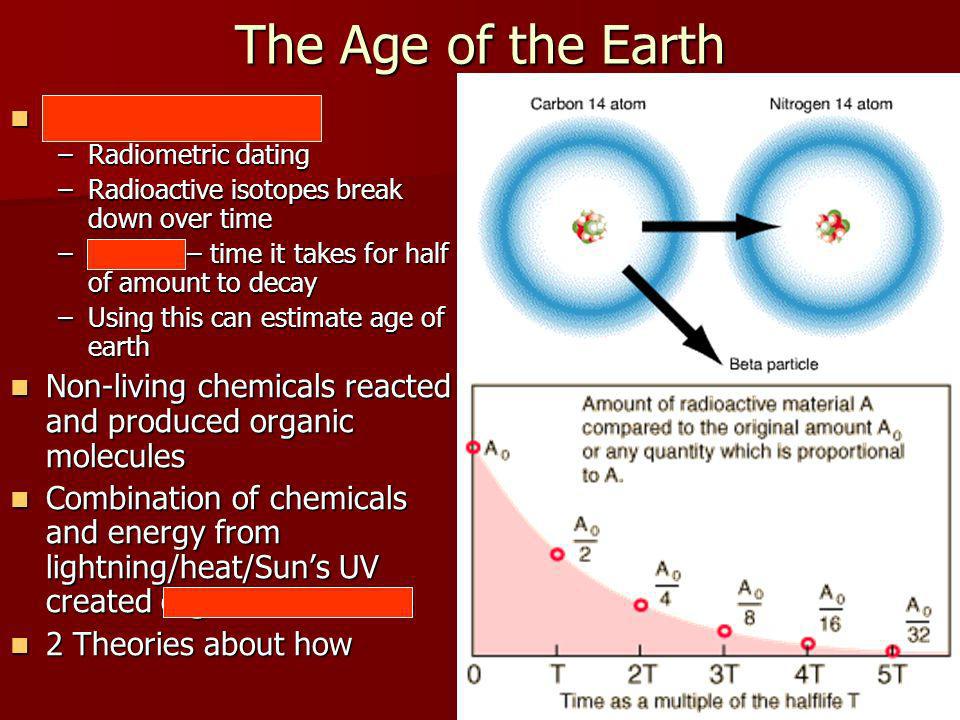
Cara Santa Maria, here. One of the ways that researchers measure the age of organic material is through carbon dating. In , Willard Libby won a Nobel Prize for developing this technology. See, all living things contain carbon, which has six protons and six neutrons, so in its typical form, we call it carbon But at any given time, there are trace amounts of carbon , or C14, in the atmosphere.
C14 is a radioactive isotope that's made when cosmic rays bombard nitrogen atoms at high altitudes, converting them to this excited form. When some living things, like plants and algae, make their own food through photosynthesis , they take in carbon dioxide from the air.
The Age of the Earth - Radiocarbom Dating as a Current Scientific Clock: Jonathan Ring
Trace amounts of C14 make up a tiny percentage of that carbon dioxide, and it's integrated into the tissues of the organism. Then creatures that can't make their own food through photosynthesis like us eat the ones that can, and that C14 is taken into our bodies as well. And because there's a constant quantity of C14 in the atmosphere, there's a constant, corresponding quantity of it in the bodies of all living things , at least while they're still alive.
That doesn't mean it's dangerous, only that it's unstable. Over time, it decays back into nitrogen. See, when an organism dies, it stops taking in carbon.
And the C14 in the organism's tissues starts to decay at a precise speed, but the amount of carbon stays the same, since it's not radioactive. We know that it takes 5, years for half of the C14 in a sample to decay. It takes another 5, years for half of what's left to decay, and so on. This is C14's half-life.
All radioactive isotopes have one. And if we compare the amount of C14 in a dead thing to the amount of regular carbon, voila!
Professor Timothy H. Heaton
We can find out how old it is. Now, some people who think that the earth is only 6, years old may base their claims on words in the Bible, not measurable evidence. Carbon dating only works for objects that are younger than about 50, years, and most rocks of interest are older than that.
Carbon dating is used by archeologists to date trees, plants, and animal remains; as well as human artifacts made from wood and leather; because these items are generally younger than 50, years.
Navigation menu
Carbon is found in different forms in the environment — mainly in the stable form of carbon and the unstable form of carbon Over time, carbon decays radioactively and turns into nitrogen. A living organism takes in both carbon and carbon from the environment in the same relative proportion that they existed naturally.
Once the organism dies, it stops replenishing its carbon supply, and the total carbon content in the organism slowly disappears. Scientists can determine how long ago an organism died by measuring how much carbon is left relative to the carbon Carbon has a half life of years, meaning that years after an organism dies, half of its carbon atoms have decayed to nitrogen atoms.